Now let's see how to make 1 N and 1M H2SO4 from 98% concentrated H2SO4 . This calculation varies with concentration of H2SO4. Because density varies with concentration of H2SO4 . Now we are having density 1.84 for 98% concentrated H2SO4. If you are going to use less concentrated H2SO4 then first find out density of it then put that value in below formula instead of 1.84 H2SO4 is liquid, so at first we need to convert H2SO4 molecular weight (gm) into volume (ml) let's use formula density = mass / volume volume = mass of HCl / density 98.08 / 1.84 V = 53.30 ml Here we are making 1M and 1N H2SO4 from 98% concentrated H2SO4. If you are going to use less concentrated H2SO4 then use that percentage value in below formula rather than 98%. Let's use formula 1M H2SO4 (98%) = 53.30 ml [ V ] 100% ...
When we see the sun in midday it looks yellow but at the sunset it looks red. do you know why.
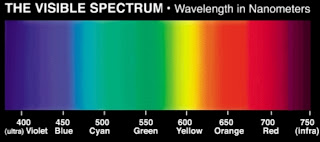
If you wants know that, you must know range of visible region.
Why sun looks yellow in midday ?
Sun looks yellow in midday because, blue, violet colours have low wavelength but high frequencies, because of that, they scatter in all direction and sky looks blue. That means we just have to consider remained yellow, orange and red colours . This three colours have low frequencies but high wavelengths, as compare to blue and violet colour . that is why they easily reach to earth surface.
There are three colours eventhough sun looks yellow because if we compare just these three colours, then we can understand orange and red colours have high wavelengths and low frequencies. But yellow colour has low wavelength and high frequency as compare to red and orange colours.
The main point is , distance between sun and earth is slightly less in the midday.
It means both red ,orange and yellow
Colours have sufficient wavelength to reach on earth surface but yellow colour has high frequency and scattering ability as compare to red and orange colours.
Just because of that high frequency and high scattering of yellow colour than red and orange colours, sun looks yellow in midday.
Why sun looks red at the sunset ?
At the sunset distance between sun and earth always greater than midday.
red colour of sun is totally depend on wavelengths of colours. Red colour has high wavelength than orange and yellow and others. Here is no matter about frequencies because in the evening distance between sun and earth is slightly more as compare to midday. Just long wavelength colour can reach to earth surface. Red and orange colours have high wavelengths that is why sun looks red and slightly orange, and Also its nearest area looks red and orange. Remained yellow colour has low wavelength but high frequency than red and orange colours hence sometimes surroundings area of sun looks slightly yellow coloured. And other colours like violet and blue are the responsible for the colour sun



Comments
Post a Comment
If you have any problems. Let me know.